Online Assessment
Instructions
1) Type in your first and last name in the
“Name” box in the top-left corner.
2) Next type in your teacher’s name in
the “ID” box.
3) Then type in your school’s name in the
“Email” box.
4) Select the best answer for each question.
5) When you
are finished click the “Grade and Submit” button.
6) The grade will be emailed
to your teacher.
|
|
|
1.
|
NOTE: If your teacher’s last
name is Elliott, then you are doing the wrong assessment.
---This assessment is only for
students of other teachers.
---If your teacher is Mr. Elliott, then please go back to the
website and click on the first assessment link instead. Thanks!
A group from
the periodic table of elements is shown below.
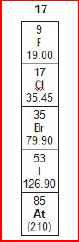
What is the atomic number of fluorine
(F)?
|
|
|
2.
|
How many total protons does each nickel atom contain, and what is the atomic
mass of Cobalt? a. | Cobalt has 28 electrons, and it is odd that
cobalt’s atomic mass is less than nickel’s mass. | b. | Cobalt has 27 electrons, and the atomic mass of Nickel is
58.7 | c. | Nickel has 28 protons, and the atomic mass of Cobalt is
58.9. | d. | Nickel has 27 electrons, and the atomic mass of
Nickel is 58.7 |
|
|
|
3.
|
Contrast atomic number and atomic
mass. a. | Atomic number is the number of neutrons in one
atom of an element.
Atomic mass is the average amount
of matter in one atom of an element | b. | Atomic
number is the number of neutrons in one atom of an element.
Atomic mass is the amount of electrons in one atom of an
element | c. | Atomic mass is the number of protons in one atom
of an element.
Atomic number is the average amount of
matter in one atom of an element. | d. | Atomic number
is the number of protons in one atom of an element.
Atomic mass is the average amount of matter in one atom of an
element |
|
|
|
4.
|
How
many protons, electrons and neutrons does each titanium atom contain? a. | Titanium atoms have 22 protons, 22 electrons and 26
neutrons. | b. | Titanium atoms have 22 protons, 22 electrons and 47.9
neutrons. | c. | Titanium atoms have 21 protons, 21 electrons and 47.9
neutrons. | d. | Titanium atoms have 22 protons, 22 electrons and 69.9
neutrons. |
|
|
|
5.
|
How many electrons and neutrons does each
neon atom contain? a. | Neon atoms have 10 electrons and 20 neutrons.
| b. | Neon atoms have 30.2 electrons and 20 neutrons.
| c. | Neon atoms have 10 electrons and 10 neutrons.
| d. | Neon atoms have 20 electrons and 10 neutrons.
|
|
|
|
6.
|
The diagram
below shows part of the periodic table of the elements.
Two-Part Question:
Which element from period 4 has the highest
atomic mass, and which element from group 17 has the lowest atomic
number?
a. | Highest atomic mass-F
(Flourine)
Low atomic
number- Kr (Krypton) | c. | High
atomic mass-K (Potassium)
Low atomic number- Ne (Neon) | b. | Highest atomic mass-Kr
(Krypton)
Lowest atomic
number-F (Flourine) | d. | High atomic mass-Fe
(Iron)
Low atomic number-
Ni (Nickle) |
|
|
|
7.
|
What is the atomic mass of an atom of
rubidium, and how many neutrons does each rubidium atom have? a. | Rubidium has 37 protons, 37 electrons, and the atomic
mass is 85.5 | b. | Rubidium has 86
neutrons, and the atomic mass is 37 | c. | Rubidium has 37
neutrons, and the atomic mass is 85.5 | d. | Rubidium has 49
neutrons, and the atomic mass is 85.5 |
|
|
|
8.
|
A group from the periodic table of elements is
shown below.
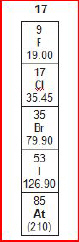
What
is the atomic mass of Bromine and the atomic number of Iodine?a. | 35 and 79.90 | c. | 79.90 and 53 | b. | 9 and
19.00 | d. | 53 and 79.90 |
|
|
|
9.
|
How many neutrons does each boron atom contain, and what is Boron’s
atomic number? a. | Boron has 16 neutrons, and the atomic number is
5. | b. | Boron has 10.8 neutrons, and the atomic number is
15.8 | c. | Boron has 5 neutrons, and the atomic number is
11. | d. | Boron has 6 neutrons, and the atomic number is
5. |
|
|
|
10.
|
The diagram
below shows part of the periodic table of the elements.
Which element
from Period 2 has the lowest atomic mass?
|