Online Assessment
Instructions
1) Type in your first and last name in the
“Name” box in the top-left corner.
2) Next type in your teacher’s name in
the “ID” box.
3) Then type in your school’s name in the
“Email” box.
4) Select the best answer for each question.
5) When you
are finished click the “Grade and Submit” button.
6) The grade will be emailed
to your teacher.
|
|
|
1.
|
NOTE: This assessment is only for Mr. Elliott’s students.
---If you are not one of Mr. Elliott’s students then please go back to the website and
click on the link for other teachers. Thanks!
Look at the diagram below of one atom of boron (B).
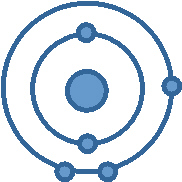
a) Based
on the number of valence electrons, what group number is boron found in?
b) Based on the number
of electons shells, what period number is boron found in? a. | Group 3, Period 3 | c. | Group
2, Period 13 | b. | Group 13, Period
2 | d. | Group 13, Period 5 |
|
|
|
2.
|
A periodic table of the elements is shown
below.
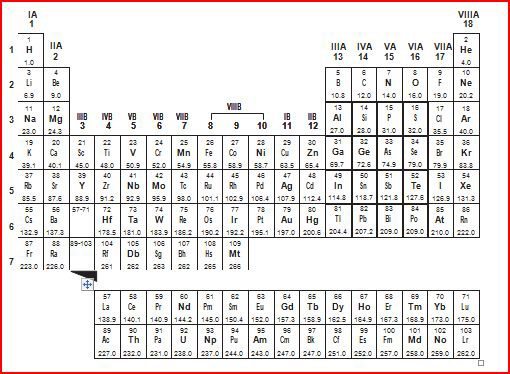
Based on their locations on the periodic table, which two elements share the
most similar chemical
properties? a. | K and
Kr | b. | H and
I | c. | Be and
Ba | d. | S and
Sn |
|
|
|
3.
|
In the periodic table shown below, some
elements are shaded.
Based on its position in the periodic table, which element is the
heaviest?a. | krypton (Kr) | b. | argon
(Ar) | c. | neon (Ne) | d. | helium
(He) |
|
|
|
4.
|
A periodic table of the elements is
shown below.
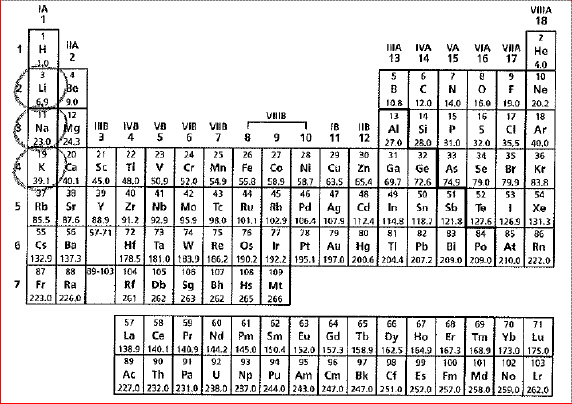 The circled elements share which
characteristic? The circled elements share which
characteristic?a. | They rarely form chemical bonds. | b. | They are alkali metals | c. | They are
nonmetals | d. | They have the same number of electrons. |
|
|
|
5.
|
A
periodic table of the elements is shown below.
What do all three of the shaded elements shown
above (Fe, Co and Ni) have in common? a. | They are all halogens with 4
electron shells. | b. | They are all poor electrical conductors. | c. | They are all metals with 4 valence
electrons. | d. | They are all metals with 4 electron
shells. |
|
|
|
6.
|
Three elements are shaded in the following
periodic table.
Nitrogen (N), Oxygen (O), and Fluorine (F)

Which statement best describes all three
shaded elements shown above? a. | They all are metals in the second
period. | b. | They all have the same atomic numbers and the same
atomic mass. | c. | They are all in
period 2, so they all have 2 electron shells. | d. | They are all in
period 4, so they all have 4 electron shells. |
|
|
|
7.
|
All three of the elements shown below are in period four.

How
many electron shells does each atom of calcium contains, and what is the atomic mass of titanium?
a. | All elements in the 4th period have 6 electron shells.
Titanium’s atomic mass is 47.9 | b. | All elements in
period 4 have 4 electron shells. Titanium’s atomic mass is 22 | c. | All elements in period 4 have 20 electron shells. Titanium’s atomic mass
is 47.9 | d. | All elements in period 4 have 4 electron shells.
Titanium’s atomic mass is 47.9 |
|
|
|
8.
|
All three of these elements are in the second
period.
12.0
C
Carbon
6 | 14.0
N
Nitrogen
7 | 15.9
O
Oxygen
8 | | | |
Two-Part Question
How many electron shells does each
atom of nitrogen contain, and what is the atomic number of carbon?
a. | They all have 2 electron shells. Carbon’s atomic
number is 6. | b. | Nitrogen has 7
electron shells. Carbon’s atomic mass is 12.0 | c. | They all have 2 electron shells. Nitrogen’s atomic number is
6. | d. | They all have 4 electron shells. Carbon’s atomic
number is 6. |
|
|
|
9.
|
The periodic table of elements is shown
below.

Which statement best describes the reactivity of the shaded elements nitrogen
(N), oxygen (O), and fluorine (F)?a. | Fluorine is the least reactive of all the shaded
elements. | b. | The nonmetals increase in reactivity from right to left
in the same period. | c. | The nonmetals
decrease in reactivity from right to left in the same period. | d. | Oxygen is the most reactive of all the elements
listed. |
|
|
|
10.
|
Two-Part Question
Which elements of the periodic table are shaded
above and what is one key property of these elements? a. | Metals, and they are good electrical
conductors. | b. | Metalloids, and
they do not conduct electricity. | c. | Lanthanides and
Actinides. They are all non-reactive noble gases | d. | Nonmetals, and they are good electrical
conductors. |
|