Online Assessment
Instructions
1) Type in your first and last name in the
“Name” box in the top-left corner.
2) Next type in your teacher’s name in
the “ID” box.
3) Then type in your school’s name in the
“Email” box.
4) Select the best answer for each question.
5) When you
are finished click the “Grade and Submit” button.
6) The grade will be emailed
to your teacher.
|
|
|
1.
|
NOTE: This assessment is only for Mr.
Elliott’s students.
---If you are not one of Mr. Elliott’s students then please go
back to the website and click on the link for other teachers. Thanks!
A student in science class combined two liquids. The temperature increased, a
foul smell was given off, and a solid substance was formed.
The student could best
classify this reaction as a...?
a. | mass change. | c. | chemical change. | b. | physical
change. | d. | weight change. |
|
|
|
2.
|
A green powder (a solid) and clear water (a liquid)
were combined together in a beaker.
After combining these two items, observations were recorded.
Which of the following observations indicates that a chemical change
occurred?
a. | The powdery substance dissolved into the clear
liquid. | b. | Nothing special was observed. The powder merely sank to
the bottom of the liquid. | c. | The combination
turned a green color. | d. | A gas was
released, a popping noise was heard, and the temperature
increased. |
|
|
|
3.
|
Which statement best describes a physical
change?
a. | An empty Coke can is crushed
flat. | b. | Wood burning on a camp fire releases gases into the
air. | c. | A copper penny exposed to the air forms rust (copper
oxide) | d. | A process separates water into hydrogen and
oxygen. |
|
|
|
4.
|
When air is heated, it expands and rises. Hot-air
balloon pilots use this phenomena to their advantage. These pilots use a gas-flame blower to increase
the temperature of the air inside the balloon making the balloon rise.
This
description of hot-air balloon operations includes a physical change example and a chemical change
example.
Select the choice below that correctly identifies
both of these changes.
a. | The heated air is a chemical change. The burning fuel is
a physical change. | b. | The balloon rising
is a chemical change due to overcoming the force of gravity. | c. | The burning fuel is a chemical change. The heated air is a physical
change. | d. | The wind blowing the balloon is a physical change. The
flame is a chemical change. |
|
|
|
5.
|
Which of the following is an example of a physical change but NOT a
chemical
change?
a. | An old car rusts away out in a grassy field. | b. | A child forgets to
put the jug of milk back in the refrigerator, and it turns sour. | c. | A wood log burns on
a camp fire. | d. | A man boils water to make hot tea. |
|
|
|
6.
|
If some rain water happens to seep down into a
crevice in a rock and it freezes, the volume of the ice will increase and it can crack the rock as it
expands. This is an example of weathering. Which of the following statements
most accurately describes this type of weathering?
a. | The rock physically changed as ice expanded inside the
crack. | b. | The rock remained unchanged during this weathering
process. | c. | A chemical change occurred as the ice cracked the rock. | d. | This is a physical change since heat was given off and gas was
released. |
|
|
|
7.
|
During phase changes, the appearance of a substance
is changed, but there is no change at the atomic level.
Which of the following is an
example of a physical change (a change of phase) but NOT a chemical change?
a. | A tree stores energy from the Sun in its fruit. | b. | A log gives off heat
and light as it burns. | c. | A water pipe freezes and cracks on a cold
night. | d. | A penny lost in the grass slowly changes color. |
|
|
|
8.
|
A group of students observed a glass of water
inside a vacuum chamber. The vacuum chamber was attached to a pump that sucked out all of the air
from the chamber. When the air was removed, the water began to boil at room temperature.
Which of the following statements BEST explains what happened in the scenario
described above?
a. | Water is naturally liquid and must undergo a chemical
change in order to boil at room temperature. | b. | When the air was
removed, the water molecules could no longer float. Therefore the liquid began to boil.
| c. | The temperature increased until the water boiled. This
is evidence of a chemical change. | d. | The water
underwent a physical change and turned into vapor when air pressure was removed
. |
|
|
|
9.
|
Copper carbonate being heated in a test
tube is shown in the diagram below.
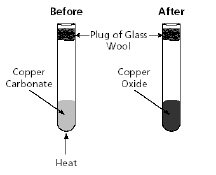 Which statement best describes what happened in
this experiment?a. | Copper oxide decomposed when heated, causing a chemical
change. | b. | Copper carbonate decomposed when heated, causing a chemical change. | c. | Copper oxide
reacted with the glass wool, causing a physical change. | d. | Copper carbonate reacted with the glass wool, causing a physical
change. |
|
|
|
10.
|
Two students combined a solid with a liquid in a container. They
observed a flash, and they saw and smelled gas being released. They also noticed that the container
grew hot to the touch.
How can they be certain that they observed a chemical change instead of
just a physical change.
a. | The saw it flash, felt the
temperature increase, and observed gas being released | b. | The can be certain because the liquid changed into
a gas (change of state) | c. | The know this was a chemical change because it changed
color. | d. | This was definitely a chemical reaction because the solid dissolved in the
liquid. |
|