Online Assessment
Instructions
1) Type in your first and last name in the
“Name” box in the top-left corner.
2) Next type in your teacher’s name in
the “ID” box.
3) Then type in your school’s full name in the
“Email” box.
4) Select the best answer for each question.
5) When you
are finished click the “Grade and Submit” button.
6) The grade will be emailed
to your teacher.
|
|
|
1.
|
NOTE: If your teacher’s last
name is Elliott, then you are doing the wrong assessment.
---This assessment is only for
students of other teachers.
---If your teacher is Mr. Elliott, then please go back to the
website and click on the first assessment link instead. Thanks!
A chart listing the density of four metals is shown below.
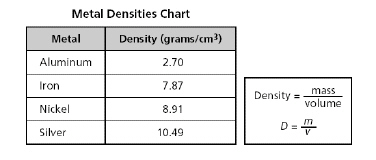
A
student determines that a metal has a volume of 3 cubic centermeters and a mass of 8.1 grams.
Using the formula above, this unknown metal is most likely...?
a. | iron | b. | aluminum | c. | nickel | d. | silver |
|
|
|
2.
|
Sample | Volume | Mass | W | 100
ml | 100 g | X | 100
ml | 80 g | Y | 100
ml | 108 g | Z | 100
ml | 90 g | | | |
The table shows the masses of different samples of liquid.
Which sample
has the GREATEST density?a. | Sample X | b. | Sample Z | c. | Sample Y | d. | Sample
W |
|
|
|
3.
|
| Sample | A | B | C | D | | Volume | 100 cc | 100 cc | 100 cc | 100 cc | | Mass | 100 g | 80
g | 108 g | 90 g | | Density | 1.6 g/cc | 0.8
g/cc | 1.03 g/cc | 1.4 g/cc | | | | | |
Three of the four
density values shown above are wrong.
Which density value
shown above is correct?a. | Sample C | b. | Sample
A | c. | Sample B | d. | Sample
D |
|
|
|
4.
|
Your science teacher conducts a density column demonstration using the following
3 liquids. | Designation | Substance | Density | | Liquid 01 | Water
| 1.0 g/mL | | Liquid
02 | Liquid Mercury | 13.6 g/mL | | Liquid 03 | Turpentine | 0.8 g/mL | | | |
He
pours each liquid into a graduated cylinder. Select the answer choice below that displays the
density of the liquid located at level B.a. | 12.4 g/cm3 | c. | 1.0 g/mL | b. | 13.6 g/mL | d. | 0.8 g/mL |
|
|
|
5.
|
A student is trying to determine the density of a
cube using the formula shown below.
What
is the density of the cube if it has a mass of 2 grams and a volume of 1 cubic
centimeter?
a. | 2.0 g/cm3 | b. | 1.0 g/cm3 | c. | 3.0
g/cm3 | d. | 0.5
g/cm3 |
|
|
|
6.
|
The equation for density is shown below.
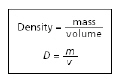 Which block has the GREATEST density?
|
|
|
7.
|
The
density for iron is 7.874 g/cm3.
What would be the MASS of a 25 cm3 piece of
iron?
HINT: MVD (mass = volume x density)
a. | 196.85g/cm3 | c. | 196.85g | b. | 393.7g | d. | 3.18g |
|
|
|
8.
|
Substance | Mass | Volume | Density | Lead | 22.72g | 2cm3 | ??? | Oak | 80g | 50cm3 | 1.6g/cm3 | Sludge | 36g | 50cm3 | ??? | | | | |
Two-part
question:
(A) What is the density of lead?
(B) What
is the density of sludge ?a. | A) Lead: 1.6g/cm3
B) Sludge: .72g/cm3
| c. | A) Sludge: 11.36g/cm3
B) Lead: .72g/cm3
| b. | A) Lead: 11.36g/cm3
B) Sludge: 1.6g/cm3
| d. | A) Lead:
11.36g/cm3
B) Sludge: .72g/cm3 |
|
|
|
9.
|
The density for aluminum is
2.7g/cm3.
What would the VOLUME be for a piece of
aluminium that has a mass of 13g?
HINT: VDM
volume = density divides the mass (mass divided by density).
a. | 4.81cm3 | c. | 4.81g/cm3 | b. | 0.207cm3 | d. | 35.1cm3 |
|
|
|
10.
|
Sample | Volume | Mass | One | 10
ml | 2 g | Two | 10
ml | 3 g | Three | 20
ml | 2 g | Four | 100
ml | 50 g | | | |
The table shows the masses of different samples of liquid.
Which sample
has the LEAST density?a. | Sample One | b. | Sample Four | c. | Sample Twp | d. | Sample
Three |
|